The Ionic Jewellery Cleaning Process
Ionic cleaning is an extremely simple yet elegant process, and the basis of all JCR systems. This process works in two simultaneous ways:
It electrochemically removes tarnish from both silver and gold by converting it into hydrogen sulphide gas. This is the gas in the atmosphere that initially caused the tarnish. More information on the tarnishing process and how to reduce minimise it…
It also removes surface grease and oil (finger prints etc.) along with the particulate matter (dirt) attached to it.
This is an extremely gentle & fast process. It is safe on all natural stones and precious metal without using any toxic or reactive chemicals. The following explains the basic science behind the principle used in all our products.
The Ionic Jewellery Cleaning Process
This is a classic and well known example of Ionic cleaning. Ionic cleaning requires a current flow between two electrodes through a conductive solution (an electrolyte). The item being cleaned is electrically attached to the cathode (rested on the aluminium foil). The electrolyte is made using hot water and backing soda. In this case the electrical energy is caused by dissimilar metals. For cleaning jewellery in particular, this method is not recommended for several reasons:
There is no way of measuring or regulating the flow of current – too much current will cause burning and erosion to the silver.
The aluminium will always dissolve into the solution (the anode is sacrificial), this will then deposit itself onto the item being cleaned. This will stain, and also later forms a type tarnish that can’t be as easily removed.
The pH of the solution will quickly drop causing the solution to become acidic – this will cause erosion and damage delicate stones.
Tap water is used. Tap water normally contains dissolved minerals and metal salts that can potentially be deposited on the item being cleaned resulting in staining.
The solution is hot. Thermal shock is particularly problematic for delicate stones.
The electrical energy flowing through the electrolyte causes hydrogen to be formed on the surface of the silver. Hydrogen is a very reactive atom. It combines immediately with the sulphur which was attached to the metal as silver sulphide creating hydrogen sulphide. Hydrogen sulphide is a gas which bubbles to the surface and can be smelt as bad eggs.
The JCR Ionic Reduction Process - Tarnish Removal
The electrochemical process described above forms the basis of the JCR Ionic reduction process. However, instead of using dissimilar metals to cause an uncontrolled current flow through the electrolyte, JCR cleaners provide a carefully regulated DC current. The cleaning current is automatically switched off at the end of the cycle, and a small bias current is maintained in order to prevent any dissolved metal salts from plating out of the solution, and onto the item being cleaned (causing staining).
The JCR electrodes are made of 316 stainless steel (the rod), and sterling silver (the hook). These metals have been specifically chosen so that they will not contaminate the electrolyte, and complicate the process.
The electrolyte (Ion-Sol) is formulated and manufactured solely by JCR Solutions specifically for cleaning delicate jewellery. It is supplied as a high purity concentrate, and is made into a working solution by diluting with distilled water. Ion-Sol contains a powerful surfactant, a wetting agent and a pH buffer to maintain a stable and slightly alkaline solution. The process is also performed at room temperature. This protects precious metal and delicate stones.
The diagram below illustrates the ionic tarnish removal process – showing the creation of hydrogen ions, and the combination of hydrogen with the silver sulphide (tarnish). This forms hydrogen sulphide, which simply bubbles out of the solution.
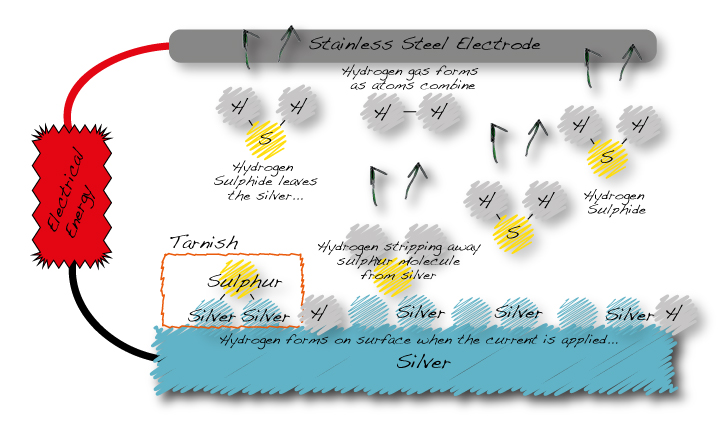
Tarnish being converted back into the gas that caused it through electrolysis
JCR Ionic Process - Grease and Oil Removal
JCR Ion-Sol electrolyte contains a carefully chosen surfactant. Surfactant molecules have a head and a tail. The tail has an affinity for oil and actively searches it out and attaches itself to it.
The head of the molecule protrudes from the surface and exhibits a polarised electrical charge. The metal onto which the oil is attached also has a charge. The charge is the same as the protruding head of the surfactant molecule. The like poles repel each other. As the surfactant molecule is firmly attached to the oil it wants to pull itself away from the surface of jewellery and towards the stainless steel electrode.
The diagram below illustrates the oil and grease emulsification and stripping process.
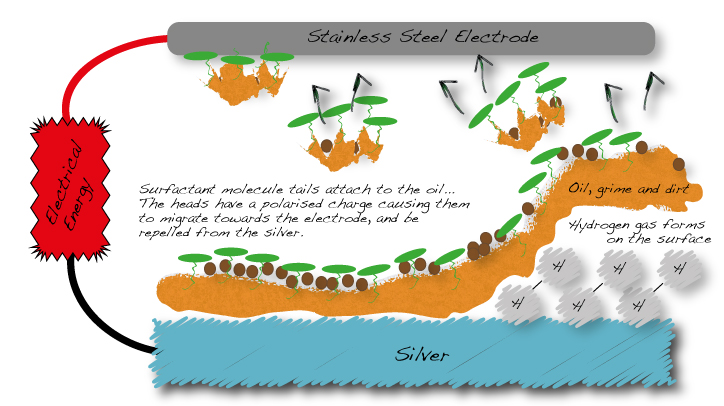
JCR Ion-Sol: electrolytic removal of grime, oil and dirt from metal with a polarised surfactant
As can be seen in the above diagram, millions of hydrogen bubbles are being formed on the surface of the silver. These have the effect of gently prying away the oil with its grime from the surface and breaking it up into small pieces. The pieces are repelled by the same charge as on the surface of the jewellery.
The oil is emulsified by the action of the surfactant preventing it from being redeposited on the surface of the metal. Non conductive surfaces of stones are cleaned in the same way as the metal. The charge of the attached surfactant heads is similar to that of the jewellery and because of their proximity to the metal they repelled away from it and towards the stainless electrode. Hydrogen bubbles help to scrub the surface and expose any remaining oil to new surfactant molecules. The whole process is so very simple, complete and extremely effective.
The removal of grease when using a JCR system is particularly striking when cleaning jewellery set with diamonds. Diamonds have a natural affinity with grease and oil. Unlike any other method of degreasing, when Ion-Sol is used, the electrical charge actively repels the surfactant and prevents the grease from reattaching to the jewellery being cleaned. This is in marked contrast to using an ultrasonic cleaner. With an ultrasonic cleaner, although the grease is very effectively removed from the jewellery, it inevitably ends up forming into a thin greasy film floating on the top of the cleaning solution. When the jewellery is removed from the ultrasonic tank, the floating grease layer redeposits itself back onto the surface of any item being cleaned…
